- Blog
- Harris Bipod For Tikka T3x
- M3u Playlist Url 2019
- Crack Pwdump Hashes
- Glm.fit: Algorithm Did Not Converge
- Perbedaan Steril Non Beta Laktam
- Adobe Creative Suite Master Collection Cs6 Keygen
- Arma Armed Assault Download
- Akademija Za Umjetnost I Kulturu
- Tenorshare 4ukey Licensed Email And Registration Code
- Notebook Lenovo G50 45 Recovery Disk Download
- Gta 5 Apk
- John Deere Lt160 Parts
- Download Directo Fortnite Pc
- Bhavan Nirman Vastu Shastra In Hindi Pdf
- Approximation Theory And Approximation Practice Ebook Download
- Pokemon Vortex Promo Codes
Center for Drug Evaluation and ResearchThis guidance describes the importance of implementing manufacturing controls to prevent cross-contamination of finished pharmaceuticals and active pharmaceutical ingredients (APIs) with non-penicillin beta-lactam drugs. This guidance also provides information regarding the relative health risk of, and the potential for, cross-reactivity in the classes of sensitizing betalactams (including both penicillins and non-penicillin beta-lactams). Finally, this guidance clarifies that manufacturers generally should utilize separate facilities for the manufacture of non-penicillin beta-lactams because those compounds pose health risks associated with crossreactivity.
Sediaan mata merupakan produk steril yang secara esensial bebas dari partikel asing, senyawa dan pengemasannya sesuai untuk pemakaian dalam mata. Ini khususnya berbahaya terhadap sediaan non steril yang dimasukkan ke dalam mata ketika kornea terluka. Aminoglikosida diinaktivasi oleh antibiotik beta laktam yang bervariasi, gentamisin.
Adults75 mg nebulized 3 times daily for 28 days then 28 days off aztreonam. Doses should be administered at least 4 hours apart. For patients taking multiple inhaled therapies, the recommended order of administration is bronchodilator, mucolytic, and then aztreonam. Patients in clinical trials were required to have been off antibiotics for at least 28 days prior to treatment with the study drug. Statistically significant improvements in respiratory symptoms were observed in both adults and pediatrics; however, these improvements were substantially smaller in adult patients. The treatment difference in the percent change in FEV1 between aztreonam and placebo at day 28 of therapy was statistically significant at 10% (95% CI, 6% to 14%). Improvements in FEV1 were comparable between adults and pediatrics.
Two weeks after completion of therapy, the difference in FEV1 between aztreonam and placebo had decreased to 6% (95% CI, 2% to 9%). Safety and efficacy have not been demonstrated in patients colonized with Burkholderia cepacia. Children and Adolescents 7 to 17 years75 mg nebulized 3 times daily for 28 days then 28 days off aztreonam. Doses should be administered at least 4 hours apart.
For patients taking multiple inhaled therapies, the recommended order of administration is bronchodilator, mucolytic, and then aztreonam. Patients in clinical trials were required to have been off antibiotics for at least 28 days prior to treatment with the study drug. Statistically significant improvements in respiratory symptoms were observed in both adults and pediatrics; however, these improvements were substantially smaller in adult patients. The treatment difference in the percent change in FEV1 between aztreonam and placebo at day 28 of therapy was statistically significant at 10% (95% CI, 6% to 14%). Improvements in FEV1 were comparable between adults and pediatrics. Two weeks after completion of therapy, the difference in FEV1 between aztreonam and placebo had decreased to 6% (95% CI, 2% to 9%).
Safety and efficacy have not been demonstrated in patients colonized with Burkholderia cepacia. Children and Adolescents 2 to 17 years150 mg/kg/day IV divided every 6 to 8 hours (Max: 8 g/day) is recommended by the Cystic Fibrosis Foundation and European Consensus Committee; 150 to 200 mg/kg/day IV divided every 6 to 8 hours (Max: 8 g/day) is recommended by the UK CF Trust Working Group. However, it has been suggested that doses of 200 to 300 mg/kg/day IV divided every 6 hours (Max: 8 to 12 g/day) may be necessary to maintain adequate serum concentrations in the treatment of pseudomonal isolates with higher MICs. The FDA-approved product labeling does not provide specific dosage recommendations, but states that doses higher than usual may be necessary for patients with cystic fibrosis. Infants and Children 9 months to 1 year150 mg/kg/day IV divided every 6 to 8 hours is recommended by the Cystic Fibrosis Foundation and European Consensus Committee; 90 to 120 mg/kg/day IV divided every 6 to 8 hours is recommended by the UK CF Trust Working Group.
However, it has been suggested that doses of 200 to 300 mg/kg/day IV divided every 6 hours may be necessary to maintain adequate serum concentrations in the treatment of pseudomonal isolates with higher MICs. The FDA-approved product labeling does not provide specific dosage recommendations, but states that doses higher than usual may be necessary for patients with cystic fibrosis. Adults1 to 2 g IV/IM every 8 to 12 hours for moderately severe systemic infections. For severe, life-threatening infections, 2 g IV every 6 to 8 hours (Max: 8 g/day).
For ICU patients with a beta-lactam allergy, guidelines recommend aztreonam in combination with a respiratory quinolone (i.e., levofloxacin, moxifloxacin). For suspected P. Aeruginosa, add ciprofloxacin or levofloxacin or azithromycin plus an aminoglycoside. In patients with risk factors for MRSA, add vancomycin or linezolid. Guidelines recommend treatment for a minimum of 5 days; the patient should be afebrile for 48 to 72 hours with no more than 1 sign of clinical instability before discontinuation. Adults2 g IV as a single dose within 60 minutes prior to the surgical incision; or alternately, 1 g IV as a single dose for gynecologic procedures. Intraoperative redosing 4 hours from the first preoperative dose and a duration of prophylaxis less than 24 hours for most procedures are recommended by clinical practice guidelines.
Clinical practice guidelines recommend aztreonam in combination with cefazolin, or for beta-lactam allergic patients, clindamycin or vancomycin, for urologic procedures involving implanted prosthesis. Aztreonam in combination with an appropriate antimicrobial with gram-positive activity (i.e., clindamycin or vancomycin) is also recommended as an alternate therapy for patients with a beta-lactam allergy undergoing gastrointestinal, biliary tract, uncomplicated appendectomy, colorectal, hysterectomy, or abdominal transplantion procedures.
Infants, Children, and Adolescents30 mg/kg/dose IV as a single dose (Max: 2 g/dose) within 60 minutes prior to the surgical incision. Intraoperative redosing 4 hours from the first preoperative dose and a duration of prophylaxis less than 24 hours for most procedures are recommended by clinical practice guidelines. Clinical practice guidelines recommend aztreonam in combination with cefazolin, or for beta-lactam allergic patients, clindamycin or vancomycin, for urologic procedures involving implanted prosthesis. Aztreonam in combination with an appropriate antimicrobial with gram-positive activity (i.e., clindamycin or vancomycin) is also recommended as an alternate therapy for patients with a beta-lactam allergy undergoing gastrointestinal, biliary tract, uncomplicated appendectomy, colorectal, or abdominal transplantion procedures.
Infants, Children, and Adolescents50 mg/kg/dose IV every 6 to 8 hours (Max: 1 to 2 g/dose), in combination with other antimicrobials, has been successfully used for the empiric treatment of febrile neutropenia in pediatric patients. Guidelines for the management of fever and neutropenia in cancer patients recommend monotherapy with an antipseudomonal beta-lactam or a carbapenem as empiric treatment in high-risk patients; addition of a second gram-negative antimicrobial agent, such as aztreonam, is recommended for patients who are clinically unstable, when a resistant infection is suspected, or for centers with high rates of resistant pathogens.†Indicates off-label use. Renal ImpairmentDosage adjustment due to renal impairment is necessary for systemic dosage only. Intravenous AdministrationPowder vials for injectionReconstitutionFor IV push, reconstitute each vial with 6 to 10 mL of Sterile Water for Injection.For intermittent IV infusion, reconstitute with at least 3 mL of Sterile Water for Injection per g of aztreonam. Intramuscular AdministrationReconstitutionReconstitute vials with at least 3 mL of an appropriate diluent per g of aztreonam.Shake immediately and vigorously.Storage: For solutions at concentrations not exceeding 20 mg/mL (2% w/v) and for solutions at concentrations exceeding 20 mg/mL (2% w/v) using Sterile Water for Injection or 0.9% Sodium Chloride Injection, use within 48 hours if kept at room temperature (15 to 30 degrees C or 59 to 98 degrees F) or within 7 days if kept under refrigeration (2 to 8 degrees C or 38 to 48 degrees F). For other solutions at concentrations exceeding 20 mg/mL (2% w/v), use promptly after preparation.44907Intramuscular injectionIn adults, inject doses of 1 g or less deeply into a large muscle (i.e., upper outer quadrant of the gluteus maximus or lateral part of the thigh).According to the FDA-approved product labeling, there are insufficient data regarding intramuscular administration of aztreonam in pediatric patients 44907; however, aztreonam has been administered IM to pediatric patients in clinical practice when acceptable IV access was not available.
Inject deeply into a large muscle mass (e.g., anterolateral thigh). When multiple IM injections are necessary, rotate administration sites.In general, IM administration of antibiotics in very low birth weight premature neonates is not practical due to small muscle mass, and absorption is unreliable due to hemodynamic instability that is relatively common in this population.53249Aztreonam is well tolerated; do not admix with any local anesthetic agent.44907.
Oral Inhalation AdministrationReconstitution of nebulized solutionDo not reconstitute aztreonam for inhalation until ready to administer a dose.Gently tap the aztreonam vial so that the powder settles to the bottom of the vial.Using the blue cap tab, slowly flip up the blue cap. Pull the blue cap down to a horizontal position, where the bottom of the blue cap faces up.Slowly pull the blue cap in a counterclockwise direction until the metal seal opens and is completely removed. Do not twist the blue cap.Carefully remove the rubber stopper.Twist the tip off of the provided 1 mL sterile diluent (0.17% Sodium Chloride) ampule and squeeze contents into the aztreonam vial.Gently swirl the vial until the contents have completely dissolved.43523Administration of nebulized solutionHave patient use a bronchodilator before administration of the aztreonam nebulized solution. A short-acting bronchodilator can be administered 15 minutes to 4 hours before aztreonam. Alternatively, a long-acting bronchodilator can be administered 30 minutes to 12 hours before aztreonam.
For patients taking multiple inhaled therapies, the recommended order of administration is bronchodilator, mucolytic, and then aztreonam.Administer aztreonam immediately after reconstitution using an Altera Nebulizer System only. Do not administer via any other nebulizer.Do not mix any with any other drugs.Administration typically takes 2 to 3 minutes via the nebulizer mouthpiece.43523. STORAGEAzactam:- Avoid excessive heat (above 104 degrees F)- Discard product if it contains particulate matter, is cloudy, or discolored- Store at controlled room temperature (between 68 and 77 degrees F)- Store in original package until time of useCayston:- Discard product if it contains particulate matter, is cloudy, or discolored- Protect from light- Reconstituted product should be used immediately. Discard unused portion- Refrigerate (between 36 and 46 degrees F)- Unrefrigerated product can be stored at temperatures not exceeding 77 degrees F for 28 days.
Carbapenem hypersensitivity, cephalosporin hypersensitivity, penicillin hypersensitivityAztreonam is a monocyclic beta-lactam. The incidence of allergic drug reactions to aztreonam is estimated at roughly 2% from reported clinical trial literature, and some reactions are consistent with IgE-mediated responses. Use would usually be contraindicated in any patient with direct aztreonam hypersensitivity.
The manufacturer states that aztreonam should be used cautiously in patients with sensitivity to any beta-lactam related antibiotic (e.g., penicillin hypersensitivity, cephalosporin hypersensitivity, carbacephem hypersensitivity, carbapenem hypersensitivity) due to the minor chemical structural similarities in the agents. However, in actuality, such cross-sensitive reactions to aztreonam are thought quite rare, and there is clinical evidence to support aztreonam's low potential for such events. Patients with known and established penicillin allergy who receive skin testing to determine if they will cross-react to aztreonam rarely have positive skin tests, and many such patients have received full therapeutic dosing with aztreonam without incident. Clinicians should be aware that it is certainly possible to have cross-hypersensitivity, but such reactions do not appear common. Renal disease, renal failure, renal impairmentAztreonam is renally excreted. Therefore, in patients with renal disease resulting in renal impairment with a glomerular filtration rate of less than 30 mL/min or renal failure requiring dialysis, systemic dosage adjustment is necessary. Since aztreonam is known to be substantially excreted by the kidney, the risk of toxic reactions to this drug may be greater in patients with impaired renal function.
Given the low systemic exposure after nebulization, clinically relevant accumulation is unlikely to occur. Nebulized aztreonam may be administered to patients with all degrees of renal impairment. Colitis, diarrhea, GI disease, inflammatory bowel disease, pseudomembranous colitis, ulcerative colitisAlmost all antibacterial agents have been associated with pseudomembranous colitis (antibiotic-associated colitis) which may range in severity from mild to life-threatening.
In the colon, overgrowth of Clostridia may exist when normal flora is altered subsequent to antibacterial administration. The toxin produced by Clostridium difficile is a primary cause of pseudomembranous colitis. It is known that systemic use of antibiotics predisposes patients to development of pseudomembranous colitis.
Consideration should be given to the diagnosis of pseudomembranous colitis in patients presenting with diarrhea following antibacterial administration. Systemic antibiotics should be prescribed with caution to patients with inflammatory bowel disease such as ulcerative colitis or other GI disease. If diarrhea develops during therapy, the drug should be discontinued. Following diagnosis of pseudomembranous colitis, therapeutic measures should be instituted. In milder cases, the colitis may respond to discontinuation of the offending agent. In moderate to severe cases, fluids and electrolytes, protein supplementation, and treatment with an antibacterial effective against Clostridium difficile may be warranted.
Products inhibiting peristalsis are contraindicated in this clinical situation. Practitioners should be aware that antibiotic-associated colitis has been observed to occur over two months or more following discontinuation of systemic antibiotic therapy; a careful medical history should be taken. Acute bronchospasmLike with other inhaled therapies, acute bronchospasm has been associated with aztreonam solution for inhalation.
Reduction of FEV1 by 15% or more immediately after administration with nebulized aztreonam was observed in 3% of patients in clinical trials despite the use of a bronchodilator prior to the administration of aztreonam. In clinical trials, patients with increased FEV1 during the 28 day treatment period were sometimes treated for pulmonary exacerbations when FEV1 declined after the 28 day treatment period. Healthcare providers should consider the patient's baseline FEV1 prior to aztreonam therapy and the presence of other symptoms when evaluating post-treatment changes in FEV1. Children, infants, neonatesAztreonam solution for inhalation is approved for use in children = 7 years old and adolescents. Safety and efficacy of nebulized aztreonam in neonates, infants and children less than 7 years old has not been established. Systemic aztreonam is approved for use in infants = 9 months, children, and adolescents.
However, systemic aztreonam is not approved in pediatric patients with septicemia and skin and skin-structure infections where the skin infection is believed or known to be due to H. Influenzae type b. Safety and efficacy of systemic aztreonam in neonates and infants less than 9 months has not been established. Breast-feedingAztreonam is excreted in breast milk at very low concentrations.
While laboratory and clinical effects in breast-fed infants are not available, the American Academy of Pediatrics (AAP) considers aztreonam to be usually compatible with breast-feeding. After a single intramuscular (IM) dose (1 gram) in 6 women, average peak milk concentrations, occurring 6 hours after the dose, were 0.3 mg/L.
After a single intravenous (IV) dose (1 gram) in 6 other women, average peak milk concentrations, occurring 2.4 hours after the dose were 0.2 mg/L. Aztreonam was detectable in milk between 2 and 8 hours after an IM dose and 1.5 and 8 hours after an IV dose. In another report, a single IV dose (1 gram) produced milk concentrations ranging 0.4—1 mg/L 1 to 5 hours after the dose with little variation in milk concentrations during this time in each woman.
On average, concentrations were slightly higher 2 hours after the dose, but in individuals the peak level occurred at various times between 1 and 4 hours. Peak plasma concentrations of aztreonam after nebulization are approximately 1% of peak concentrations observed after IV administration (500 mg). The manufacturer states that the use of nebulized aztreonam is unlikely to pose risk to the breast-feeding infant. GeriatricClinical studies of systemic and nebulized aztreonam in geriatric patients have not included sufficient numbers of patients over 65 years of age to determine if they respond differently to treatment than younger adults.
However, clinical experience has not identified differences between older and younger patients. In general, systemic aztreonam dosage selection for elderly patients should be cautious, starting at the lower end of the dosage range, reflecting the greater frequency of decreased hepatic, renal or cardiac function and concomitant disease. Because elderly patients are more likely to have decreased renal function that will decrease aztreonam elimination, renal function should be monitored;systemic dosage adjustments made accordingly. The federal Omnibus Budget Reconciliation Act (OBRA) regulates medication use in residents of long-term care facilities. According to OBRA, use of antibiotics should be limited to confirmed or suspected bacterial infections.
Antibiotics are non-selective and may result in the eradication of beneficial microorganisms while promoting the emergence of undesired ones, causing secondary infections such as oral thrush, colitis, or vaginitis. Any antibiotic may cause diarrhea, nausea, vomiting, anorexia, and hypersensitivity reactions. DRUG INTERACTIONSOral Contraceptives: (Moderate) It would be prudent to recommend alternative or additional contraception when oral contraceptives (OCs) are used in conjunction with antibiotics. It was previously thought that antibiotics may decrease the effectiveness of OCs containing estrogens due to stimulation of metabolism or a reduction in enterohepatic circulation via changes in GI flora. One retrospective study reviewed the literature to determine the effects of oral antibiotics on the pharmacokinetics of contraceptive estrogens and progestins, and also examined clinical studies in which the incidence of pregnancy with OCs and antibiotics was reported. It was concluded that the antibiotics ampicillin, ciprofloxacin, clarithromycin, doxycycline, metronidazole, ofloxacin, roxithromycin, temafloxacin, and tetracycline did not alter plasma concentrations of OCs. Antituberculous drugs (e.g., rifampin) were the only agents associated with OC failure and pregnancy.
Based on the study results, these authors recommended that back-up contraception may not be necessary if OCs are used reliably during oral antibiotic use. Another review concurred with these data, but noted that individual patients have been identified who experienced significant decreases in plasma concentrations of combined OC components and who appeared to ovulate; the agents most often associated with these changes were rifampin, tetracyclines, and penicillin derivatives. These authors concluded that because females most at risk for OC failure or noncompliance may not be easily identified and the true incidence of such events may be under-reported, and given the serious consequence of unwanted pregnancy, that recommending an additional method of contraception during short-term antibiotic use may be justified.
During long-term antibiotic administration, the risk for drug interaction with OCs is less clear, but alternative or additional contraception may be advisable in selected circumstances. Data regarding progestin-only contraceptives or for newer combined contraceptive deliveries (e.g., patches, rings) are not available.Sodium picosulfate; Magnesium oxide; Anhydrous citric acid: (Major) Prior or concomitant use of antibiotics with sodium picosulfate; magnesium oxide; anhydrous citric acid may reduce efficacy of the bowel preparation as conversion of sodium picosulfate to its active metabolite bis-(p-hydroxy-phenyl)-pyridyl-2-methane (BHPM) is mediated by colonic bacteria. If possible, avoid coadministration. Certain antibiotics (i.e., tetracyclines and quinolones) may chelate with the magnesium in sodium picosulfate; magnesium oxide; anhydrous citric acid solution. Therefore, these antibiotics should be taken at least 2 hours before and not less than 6 hours after the administration of sodium picosulfate; magnesium oxide; anhydrous citric acid solution.Warfarin: (Moderate) The concomitant use of warfarin with many classes of antibiotics, including aztreonam, may result in an increased INR thereby potentiating the risk for bleeding. Inhibition of vitamin K synthesis due to alterations in the intestinal flora may be a mechanism; however, concurrent infection is also a potential risk factor for elevated INR.
Monitor patients for signs and symptoms of bleeding. Additionally, increased monitoring of the INR, especially during initiation and upon discontinuation of the antibiotic, may be necessary. PregnancyAztreonam is classified as FDA pregnancy risk category B. There have been no adequate, well-controlled studies of aztreonam in pregnant women and it is, therefore, recommended during pregnancy only if clearly needed. Aztreonam does cross the placenta and enter fetal circulation. Systemic exposure after nebulized aztreonam is minimal.Aztreonam is excreted in breast milk at very low concentrations. While laboratory and clinical effects in breast-fed infants are not available, the American Academy of Pediatrics (AAP) considers aztreonam to be usually compatible with breast-feeding.
After a single intramuscular (IM) dose (1 gram) in 6 women, average peak milk concentrations, occurring 6 hours after the dose, were 0.3 mg/L. After a single intravenous (IV) dose (1 gram) in 6 other women, average peak milk concentrations, occurring 2.4 hours after the dose were 0.2 mg/L.
Aztreonam was detectable in milk between 2 and 8 hours after an IM dose and 1.5 and 8 hours after an IV dose. In another report, a single IV dose (1 gram) produced milk concentrations ranging 0.4—1 mg/L 1 to 5 hours after the dose with little variation in milk concentrations during this time in each woman. On average, concentrations were slightly higher 2 hours after the dose, but in individuals the peak level occurred at various times between 1 and 4 hours. Peak plasma concentrations of aztreonam after nebulization are approximately 1% of peak concentrations observed after IV administration (500 mg).
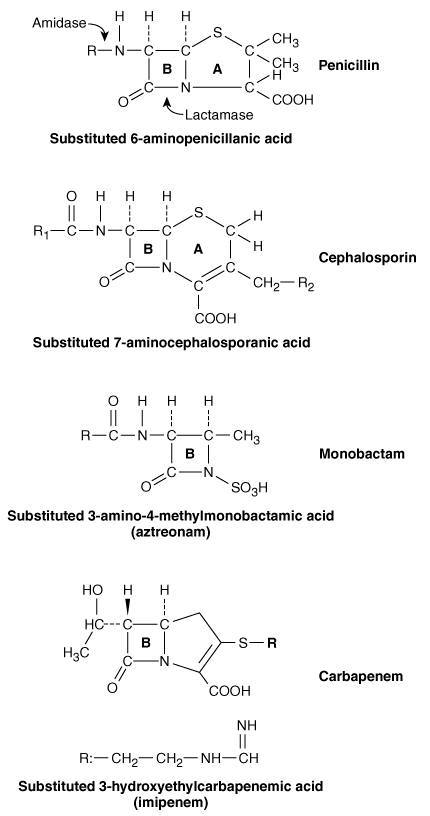
The manufacturer states that the use of nebulized aztreonam is unlikely to pose risk to the breast-feeding infant. MECHANISM OF ACTIONUnlike the penicillins and cephalosporins, aztreonam is a monobactam.
It contains a sulfonic acid group that gives the beta-lactam of aztreonam its activity. Like the penicillins and cephalosporins, aztreonam is mainly bactericidal and inhibits the third and final stage of bacterial cell wall synthesis by preferentially binding to specific penicillin-binding proteins (PBPs) that are located inside the bacterial cell wall. Penicillin-binding proteins are responsible for several steps in the synthesis of the cell wall and are found in quantities of several hundred to several thousand molecules per bacterial cell.
Penicillin-binding proteins vary among different bacterial species. In particular, aztreonam preferentially binds to PBP-3 of the gram-negative rods, and the sulfonic acid group assists in the acetylation of PBP-3. Since PBP-3 is responsible for formation of the septum during cell division, aztreonam's inhibition of these proteins causes elongation of the bacteria, inhibition of bacterial cell division, and breakage of the cell wall, which results in cell lysis and death. Aztreonam has no affinity for the PBPs of gram-positive organisms and poor affinity for anaerobic PBPs. In vitro, aztreonam has little ability to induce chromosomally mediated beta-lactamase production, although the selection of preexisting resistant organisms is still possible during clinical use.23602 31042 50323The interpretive breakpoint criteria define MICs for systemic aztreonam for Enterobacteriaceae as susceptible at 4 mcg/mL or less, intermediate at 8 mcg/mL, and resistant at 16 mcg/mL or more.
The interpretive breakpoint criteria define MICs for P. Aeurginosa as susceptible at 8 mcg/mL or less, intermediate at 16 mcg/mL, and resistant at 32 mcg/mL or more (breakpoints based on a dose of 1 g IV every 6 hours or 2 g IV every 8 hours).
The interpretive breakpoint criteria define MICs for H. Influenzae and H. Parainfluenzae as susceptible at 2 mcg/mL or less.63320 63321Bacterial resistance to aztreonam occurs via hydrolysis by beta-lactamase, alteration of the PBP, and decreased intracellular permeability.31042. PHARMACOKINETICSAztreonam is administered via nebulization, intravenously, and intramuscularly. Approximately 56—65% of aztreonam is protein-bound. Systemic aztreonam is distributed into most body tissues and fluids including lungs, liver, kidneys, bone, uterus, ovary, intestine, saliva, sputum, bile, as well as peritoneal, pleural, pericardial, and synovial fluids.
It reaches high enough concentrations within CSF to inhibit most Enterobacteriaceae when administered systemically, although it is not indicated for the treatment of meningitis. It also crosses the placenta.Hepatic metabolism is a minor pathway of excretion. Aztreonam and the inactive metabolites are excreted primarily into the urine via tubular secretion and glomerular filtration.
A small percentage is excreted in feces. The drug is also excreted in breast milk. The elimination half-life of aztreonam is 1.7 hours after systemic administration and 2.1 hours after nebulization in patients with normal renal function. Concomitant administration of probenecid or furosemide and aztreonam does not result in clinically significant increases in aztreonam serum concentrations.
Intravenous RouteAfter single aztreonam 500—1000 mg IV doses, the serum levels exceeded the MIC90 for Neisseria sp, H. Influenzae, most genera of Enterobacteriaceae and 80% of Enterobacter sp. Aeruginosa, a single 2 g IV dose of aztreonam will maintain levels exceeding the MIC90 for 4—6 hours. The same doses of aztreonam result in urine concentrations of aztreonam that exceed the MIC90 for these organisms for up to 12 hours. Approximately 60—70% of an IV or IM dose of aztreonam is recovered in the urine in 8 hours. Urinary excretion of a single dose is essentially complete 12 hours after the injection. Intramuscular RoutePeak plasma levels of aztreonam occur within 60 minutes after an IM dose.
Serum concentrations are comparable between IM and IV dosing at 1 hour (1.5 hours from the start of the IV infusion) after the dose. Following single aztreonam 500—1000 mg IM doses, the serum levels exceeded the MIC90 for Neisseria sp, H. Influenzae, most genera of Enterobacteriaceae, and 80% of Enterobacter sp.
The same doses of aztreonam result in urine concentrations of aztreonam that exceed the MIC90 for these organisms for up to 12 hours. Approximately 60—70% of an IV or IM dose of aztreonam is recovered in the urine in 8 hours. Urinary excretion of a single dose is essentially complete 12 hours after the injection. Inhalation RouteSputum and plasma concentrations exhibited considerable variability between patients receiving nebulized aztreonam in clinical trials. The mean sputum concentration 10 minutes after the first dose of nebulized aztreonam (75 mg) was 726 mcg/g. In patients receiving nebulized aztreonam 3 times daily, mean sputum concentrations 10 minutes after dose administration on days 0, 14, and 28 were 984 mcg/g, 793 mcg/g, and 715 mcg/g, respectively, indicating no drug accumulation.
The mean peak plasma concentration one hour after the first dose of nebulized aztreonam was 0.59 mcg/mL. Mean peak plasma concentrations in patients receiving nebulized aztreonam 3 times daily were 0.55 mcg/mL, 0.67 mcg/mL, and 0.65 mcg/mL on days 0, 14, and 28, respectively.
These are low systemic concentrations compared to peak serum concentrations after an IV dose (approximately 54 mcg/mL after a 500 mg dose). Evaluation of plasma and urine concentrations indicates a low systemic absorption of nebulized aztreonam. Approximately 10% of the total nebulized dose is excreted in the urine as unchanged drug compared to 60—65% after IV administration. The elimination half-life of nebulized aztreonam is 2.1 hours.
- Blog
- Harris Bipod For Tikka T3x
- M3u Playlist Url 2019
- Crack Pwdump Hashes
- Glm.fit: Algorithm Did Not Converge
- Perbedaan Steril Non Beta Laktam
- Adobe Creative Suite Master Collection Cs6 Keygen
- Arma Armed Assault Download
- Akademija Za Umjetnost I Kulturu
- Tenorshare 4ukey Licensed Email And Registration Code
- Notebook Lenovo G50 45 Recovery Disk Download
- Gta 5 Apk
- John Deere Lt160 Parts
- Download Directo Fortnite Pc
- Bhavan Nirman Vastu Shastra In Hindi Pdf
- Approximation Theory And Approximation Practice Ebook Download
- Pokemon Vortex Promo Codes
